Oxytocin and its cognate receptor have been proposed as essential mediators of social behaviors, physiological functions, and emotional responses in many animal species. Oxytocin is an evolutionarily conserved neuropeptide consisting of just nine amino acids, which is produced by a few discrete populations of neurons in the hypothalamus. In contrast, the receptors for oxytocin are expressed broadly throughout the brain and body, providing many targets for oxytocin neuromodulation.
Understanding the precise mechanisms by which this small molecule influences behavior presents a challenging endeavor. Indeed, studies of genetic models of mice deficient in oxytocin or oxytocin receptors have shown that oxytocin influences a myriad of social behaviors, including recognition of familiar and novel mouse encounters, parental care, social interactions of female with male mice during the estrous cycle, aggression, and anxiety-related behaviors in male mice. In addition to its functional diversity, oxytocin can be released from the axons, dendrites, and cell bodies of oxytocin-expressing neurons, and reaches distant targets via diffusion, adding both temporal and spatial complexities in unraveling the mechanism of oxytocin function.
In our study (PDF), e investigated how oxytocin regulates social interactions between mice using conditional genetics. In previously published work we had demonstrated that stereotypic social responses in mice is triggered by sex-specific pheromones detected by sensory neurons in the vomeronasal organ. These signals are subsequently processed in the accessory olfactory bulb and the medial amygdala before reaching hypothalamic areas controlling innate behavioral responses. Here, we set out to dissect how oxytocin influences innate behaviors triggered by pheromones, and how oxytocin signaling affects the processing of these socially relevant cues along the vomeronasal pathway.
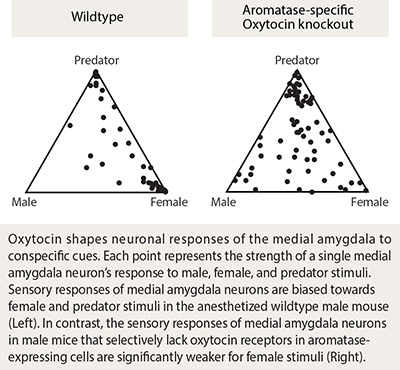
We first investigated the effect of oxytocin on the innate preference of male mice to interact with a female rather than another male. We found that a male mouse lacking oxytocin does not display the typical preference to explore a female rather than a male, and has an impaired ability to discriminate female vs. male pheromones. To identify the neural substrate of oxytocin function in sex-discrimination, we conducted genetic ablation as well as virus-mediated manipulation of oxytocin receptors, and identified a critical role of oxytocin signaling in a sub-population of medial amygdala neurons expressing the enzyme aromatase. Using multisite extracellular recordings in the medial amygdala of male mice, chemogenetics, and pharmacology, we showed that the representation of social stimuli in the medial amygdala depends on oxytocin in a manner that echoed the observed changes in behavioral sex discrimination.
This study provides significant new insights into the role of oxytocin signaling in the control of social behavior. Acting at the level of sensory-cue processing, oxytocin can participate in diverse social responses within a given animal species. Acute modulation by oxytocin provides a mechanism to fine-tune the functional configurations of neural circuits and adapt the resulting behaviors on a moment to moment basis, while maintaining the integrity of circuit connectivity.
by Shenqin Yao, Joe Bergan and Catherine Dulac


