In the brain, the function of a nerve cell (a neuron) is based both on the input it receives, via synapses onto its dendrites from upstream neurons, and the output it conveys, via synapses from its axon to downstream neurons.
However, getting a complete list of all the input and output for one neuron has not been possible until recently. The advent of methods for generating stacks of serial electron microscopy images of large volumes allows us to trace out all these connections.
In this work we make the largest volume to date of brain tissue to study the input and output of one inhibitory interneuron in the mouse visual thalamus. The visual thalamus links the retina to the visual cortex. We generated a 100 TB data set covering much of the visual thalamus and then traced out all the hundreds of inputs and outputs to one individual local inhibitory neuron.
This inhibitory neuron defied easy categorization because, in distinction to most neurons, all but one of its branches had both axonal (output) and dendritic (input) synapses.
In addition, this cell extended branches throughout much of the thalamus so that it participated in visual processing over much of the visual field and probably participated in several different kinds of visual functions (e.g., tracking moving objects and determining the shape of an object). It weakly interacted with many downstream neurons by contacting each with small numbers of synapses.
At the same time, some of its branches strongly interacted with a smaller number of neurons by establishing many synapses with each of them.
This inhibitory neuron’s diverse range of synaptic relationships allows it to participate in a mix of global and local processing but defies assigning it a single role in neural circuits.
This neuron therefore undercuts a fundamental tenet of Cajal’s “neuron doctrine” in that, rather showing that the neuron is the unit of organization, one neuron may participate in many different functions and these are spatially distributed among its many far-flung branches.
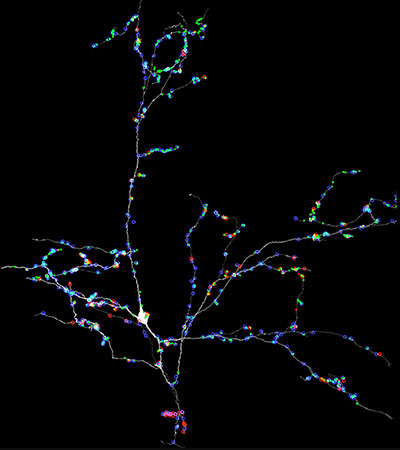
Local inhibitory neuron in visual thalamus
by Josh Morgan and Jeff Lichtman





