Parenting is absolutely fundamental for the survival and wellbeing of offspring in many species. This involves numerous efforts and sacrifices (as most parents will readily acknowledge) but does not provide immediate benefits to the caregiver. It is therefore likely that parenting relies on evolutionarily shaped, hard-wired brain circuits that coordinate its motor, motivational, hormonal and social aspects. Despite their importance, how such multicomponent behaviors are controlled at the level of neural circuits in the brains has remained largely unclear.
We previously identified a population of neurons that controls parenting in both male and female mice. These neurons, expressing the neuropeptide Galanin (Gal), are located in the medial preoptic area (MPOA) of the hypothalamus, an evolutionarily old part of the brain. Harnessing the power of mouse genetics, we now asked how this tiny set of genetically specified neurons (~0.0001% of neurons in the mouse brain) is able to control the diverse components of a complex social behavior.
We first aimed to uncover the anatomical logic of the circuits into which MPOAGal neurons are embedded. Using viruses encoding fluorescent markers, we visualized both the direct inputs into MPOAGal neurons as well as their projections to downstream brain areas. This revealed a surprising complexity: MPOAGal neurons receive inputs from more than 20 areas and send projections to most of the same areas, too. Using more sophisticated tracing methods, we uncovered a circuit architecture in which broad sets of inputs converge onto largely separate subpopulations (pools) of MPOAGal neurons that each project to a different downstream area (see Figure).
The next question was whether these distinct pools are active during discrete episodes of parenting. We first used genetically encoded calcium reporters to image the activity of all MPOAGal neurons in freely moving mice during parenting. The MPOAGal population was active during all steps of parental behavior but when we recorded from specific pools, we excitingly found evidence for differential tuning: while one pool was active only during pup grooming, a second one showed weak activation when the animal entered the nest. A third pool was active during essentially all aspects of parenting. Different pools might therefore indeed govern different aspects of parenting.
In a last set of experiments, we directly tested this by optogenetically activating or silencing individual pools. We showed that MPOAGal pools indeed mediate crucial, non-overlapping aspects of parenting (see Figure).
Our study provides a new model for how discrete elements of a social behavior are generated at the circuit level. Intriguingly, the organization uncovered here is reminiscent of the control of motor sequences by pools of neurons in the spinal cord. It will be exciting to see whether other behaviors rely on similar functional circuit architecture.
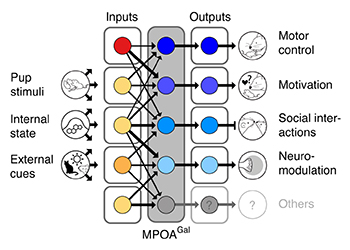
Functional architecture of the MPOAGal circuit. Broad, state- and sex-specifically activated inputs converge onto largely non-overlapping, projection-defined MPOAGal subpopulations that elicit specific aspects of parental behavior.
by Johannes Kohl and Catherine Dulac








